Chemical composition of enamel - organic and inorganic components
Enamel is the outer protective layer that covers each tooth. Dentin is hidden under it, and under it is the pulp chamber with a neurovascular bundle, pulp. In this case, the enamel layer covers only the crown (the visible part of the tooth), which passes into the neck, and that into the root. The surface of the root system is covered with cement - a solid mixture of collagen and calcium fibers. We can say that this is the dentogingival connection, because with its help the root is securely attached to the alveolus.
The top layer of teeth is considered the hardest tissue in the human body, which is due to the presence of mineral components of tooth enamel in its composition. It contains mainly calcium phosphate in the form of hydroxyapatite crystals - approximately 95-97%. The remaining part is accounted for by organic substances, and this is only 1-2%. The rest is water, approximately 2-3%.
Chemical composition of tooth enamel
The most durable are the surface layers, especially on the cutting edges and chewing surfaces of the incisors, canines and molars. Closer to the cervical region, the degree of tissue hardness decreases. At the same time, the thickness of this layer is not evenly distributed throughout the entire crown. So, for example, on the chewing surface it can be 1.5-1.6 mm, but on the sides and at the very base the enamel will be much thinner.
Main functions
The main functional task of this part of the tooth is the reliable protection of its internal structures - dentin and pulp. It is a barrier that prevents irritating mechanical, thermal and chemical effects on more sensitive and susceptible tissues. It is thanks to its hardness that we are able to bite and chew food freely and painlessly.
Generally speaking, teeth also help us reproduce sounds and, accordingly, speak. They are also responsible for the attractiveness of our smile and appearance in general, which ensures our comfort both psychologically and socially.
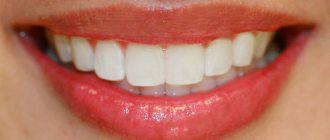
Enamel helps you bite and chew food
Anatomical and histological structure of the enamel layer
The structure of tooth enamel includes several components. Among them, two main structural units can be distinguished - enamel prisms and interprismatic substance. Let's look at them in a little more detail.
Composition of enamel prisms
The histological structure of tooth enamel provides for the presence of prisms, which consist of special enamel-forming cells called ameloblasts. And the main distinguishing feature of these prisms is the fact that they continuously intersect the surface of the enamel throughout its entire thickness and are in a position perpendicular to the junction of enamel and dentin.
Interprismatic fabric
The part of histology that studies the structure of teeth also highlights interprismatic tissue. The main difference between this component and a prism is the direction of its crystals. Enamel tufts and plates pass through the thickness of the coating and represent hypomineralized zones. The plates, called lapellae, are peculiar defects that consist mainly of organic compounds. These, in turn, can facilitate the penetration of bacteria into the protective layer of teeth and thereby help the development of carious processes1.
The photo shows a diagram of the structure of tooth enamel
It should also be noted that processes of odontoblasts are also present in the interprismatic space. They largely determine the sensitivity of the enamel. The bottom line is that the bodies of odontoblasts are located in the pulp, where the nerve endings are concentrated. In this case, the processes of these cells are located directly in the enamel.
Susceptibility of enamel to carious processes
Despite its hardness, the protective layer of our teeth still remains quite susceptible to some pathological processes, in particular, to the development of caries. This disease is a consequence of the active reproduction and spread of bacteria, the waste products of which have a destructive effect on hard tissues.
Susceptibility to such processes may be due to various factors. Among them, experts in the field of dentistry highlight the underdevelopment of the enamel coating, which is an anomaly in the formation of the dental system. Caries also often results from poor nutrition, excessive consumption of foods rich in simple carbohydrates and sugars, and lack of vitamins and beneficial microelements in the diet.
Possible prerequisites for the development of carious processes also include insufficient hygiene, the formation of abundant plaque and deposits, internal systemic pathologies leading to changes in the composition of saliva and ph balance in the oral cavity.
PERIODONTAL
A major role in the aging of periodontal tissues is played by changes in blood vessels, collagen, enzyme activity, and immunobiological reactivity, when the processes of cell decay begin to prevail over the processes of their restoration. Metabolism slows down, oxygen supply to tissues decreases, and their dehydration increases. The composition of cellular elements changes and the level of lysozyme in the gum tissue decreases.
Therefore, the treatment of periodontal diseases always includes vitamin therapy, vascular and immunostimulating drugs, and all types of massage. It is absolutely necessary to strengthen the walls of blood vessels and strengthen the immune system constantly!
What pathological processes are the upper dental layer susceptible to?
To keep your teeth healthy and your smile snow-white and radiant, it is important to maintain hygiene and respond in a timely manner to any pathological changes occurring in the oral cavity. Unfortunately, most patients seek dental care in the later stages of the disease. Let's look at what possible problems can affect the condition of the enamel and lead to undesirable consequences.
Destruction by carious and non-carious processes
The cause of destruction of hard tissues can be both carious and non-carious processes. And if with the former everything is more or less clear, then the destruction of a non-carious nature requires a thorough examination of the body for serious systemic disorders.
Caries destroys tooth enamel
Side failures can occur both in the already formed body of an adult, and in a child at the stage of eruption of milk or permanent teeth, and even in a fetus at the stage of intrauterine development. In the latter case, the problem can be triggered by illness of the expectant mother or taking serious medications while carrying the baby. However, the weakening of enamel may also be associated with hormonal changes; for example, during pregnancy, many women complain of hyperesthesia.
Pathological abrasion
The prerequisites for this problem are often pathologies of the dental system, including various forms of malocclusion or crooked position of the teeth. However, the causes can also be neurological in nature, and this is often observed in childhood.
For example, some children grind their teeth loudly during sleep, which occurs due to an unconscious but very strong clenching of the jaws. In such cases, the child is usually referred to a neurologist, prescribed a course of sedatives and an individual mouthguard to be worn at night - it will protect the teeth from abrasion.
This is what tooth enamel wear looks like
“My child was diagnosed with increased abrasion and bruxism. This is when children grind their teeth loudly at night. There is a rumor that worms are to blame for everything, but this is complete nonsense. I worm him every year as expected. The doctor said that it could be nervous and prescribed special mouth guards. Now he sleeps with them, although at first he was capricious, didn’t want to put them on, saying they were in the way. Well, what can you do, you can’t walk around without teeth!”
Lyudmila, from correspondence on the woman.ru forum
Wedge-shaped defect
This pathology manifests itself as a V-shaped connection between the tooth and the gum. The defect occurs as a result of damage to the mucosa or drooping of its edge, and there are also many reasons for this phenomenon. This could be an incorrectly selected brush, an abnormal bite, a lack of micronutrients in the body, or excessive deposits in the subgingival area as a result of insufficient oral hygiene.
This is what a wedge-shaped defect looks like
Hypo- and hyperplasia
Hypoplasia is expressed in the partial absence of enamel in different parts of the tooth or its gradual thinning against the background of serious systemic disorders in the body. Hyperplasia is the opposite phenomenon, which is characterized by excessive formation of enamel tissue and heterogeneity in the structure of the outer shell of the teeth.
The photo shows tooth enamel hyperplasia
Erosive lesion
The pathology is very common, but its causes are still poorly understood. It is rarely possible to detect it in the early stages, and most often patients seek dental care when the enamel is already completely covered with erosive spots and does not cope well with its direct protective functions.
The photo shows erosion of tooth enamel
The presumable reasons for the development of such a pathological process are endocrine disorders and the use of potent medications, leading to demineralization and destruction of the tooth surface.
Biochemistry of the oral cavity
Biochemistry of hard dental tissues
Such tissues include enamel, dentin, and cementum. These tissues differ from each other by their different origins in ontogenesis. Therefore, they differ in chemical structure and composition. And also by the nature of metabolism. In them, the enamel is of eptodermal origin, and the bone, cement, dentin are of mesentimal origin, but despite this, all these tissues have much in common, they consist of an intercellular substance or matrix of carbohydrate-protein nature and a large amount of minerals, mainly , represented by apatite crystals.
Degree of mineralization:
Enamel -> dentin -> cement -> bone.
These tissues contain the following percentages:
Minerals: Enamel-95%; Dentin-70%; Cement-50%; Bone-45%
Organic substances: Enamel-1 – 1.5%; Dentin-20%; Cement-27%; Bone-30%
Water: Enamel-30%; Dentin-4%; Cement-13%; Bone-25%.
These crystals have a hexogenal shape.
Mineral components of enamel
They are presented in the form of compounds having a crystal lattice
A(BO)K
A = Ca, Ba, cadmium, strontium
B = PO, Si, As, CO.
K = OH, Br, J, Cl.
1) hydroxyapatite - Ca (PO) (OH) in tooth enamel 75% HAP - the most common in mineralized tissues
2) carbonate apatite - CAP - 19% Ca (PO) CO - soft, easily soluble in weak acids, whole, easily destroyed
3) chlorapatite Ca (PO) Cl 4.4% soft
4) strontium apatite (SAP) Ca Sr (PO) - 0.9% is not common in mineral tissues and is common in inanimate nature.
Min. ingredients 1 – 2% in non-apatite form, in the form of calcium phosphate, dicalciferate, orthocalciphosphate. The Ca/P ratio of 1.67 corresponds to the ideal ratio, but Ca ions can be replaced by chemical elements Ba, Cr, and Mg that are similar in properties. At the same time, the ratio of Ca to P decreases, it decreases to 1.33%, the properties of this apatite change, and the resistance of the enamel to adverse conditions decreases. As a result of the replacement of hydroxyl groups with fluorine, fluorapatite is formed, which is superior in both strength and acid resistance to HAP.
Ca (PO) (OH) + F = Ca (PO) FOH hydroxyfluorapatite
Ca(PO)(OH) + 2F = Ca(PO)F fluorapatite
Ca (PO) (OH) + 20F = 10CaF + 6PO + 2OH Ca fluoride.
CaF is durable, hard, and easily leached. If the pH shifts to the alkaline side, tooth enamel destruction, enamel mottling, and fluorosis occurs.
Strontium apatite - in the bones and teeth of animals and people living in regions with a high content of radioactive strontium, they have increased fragility. Bones and teeth become brittle, strontium rickets develops, causeless, multiple bone fractures. Unlike ordinary rickets, strontium rickets is not treated with vitamin D.
Features of the crystal structure
The most typical is the hexogenal form of HAp, but there may be rod-shaped, acicular, or diamond-shaped crystals. All of them are ordered, of a certain shape, have ordered enamel prisms - this is the structural unit of enamel.
4 structures:
a crystal consists of elementary units or cells; there can be up to 2 thousand such cells. Mol.mass = 1000. A cell is a 1st order structure, the crystal itself has Mr = 2,000,000, it has 2,000 cells. Crystal is a 2nd order structure.
Enamel prisms are a 3rd order structure. In turn, enamel prisms are collected in bundles, this is a 4th order structure, around each crystal there is a hydration shell, any penetration of substances onto the surface or inside the crystal is bound in this hydration shell.
It is a layer of water associated with a crystal in which ion exchange occurs, it ensures the constancy of the enamel composition, called enamel lymph.
Water is intracrystalline; the physiological properties of enamel and some chemical properties, solubility, and permeability depend on it.
Type: water bound to enamel proteins. In the structure of HAP, the Ca/P ratio is 1.67. But there are HAPs in which this ratio ranges from 1.33 to 2.
Ca ions in HAP can be replaced by other chemical elements similar in properties to Ca. These are Ba, Mg, Sr, less often Na, K, Mg, Zn, HO ion. Such substitutions are called isomorphic, as a result the Ca / P ratio decreases. Thus, it is formed from HAP - HFA.
Phosphates can be replaced by the PO ion HPO citrate.
Hydroxites are replaced by Cl, Br, F, J.
Such isomorphic substitutions lead to changes in the properties of apatites - the resistance of enamel to acids and to caries decreases.
There are other reasons for changes in the composition of HAP, the presence of vacant places in the crystal lattice, which must be replaced with one of the ions, vacant places arise most often under the action of acids, already in the formed HAP crystal, the formation of vacant places leads to a change in the strength of the enamel, permeability , solubility, adsorptive properties.
The balance between the process of de- and remineralization is disrupted. Optimal conditions for chemicals arise. reactions on the enamel surface.
Physicochemical properties of apatite crystal
One of the most important properties of a crystal is charge. If there are 10 remaining Ca in a HAP crystal, then consider 2 x 10 = 3 x 6 + 1 x 2 = 20 + 20 = 0.
HAP is electrically neutral; if the HAP structure contains 8 Ca – Ca (PO) ions, then 2 x 8 20 = 16 < 20, the crystal acquires a negative charge. It can also be positively charged. Such crystals become unstable. They are reactive and surface electrochemical imbalance occurs. ions are in a hydration shell. They can neutralize the charge on the surface of apatite and such a crystal again becomes stable.
Stages of penetration of substances into a HAP crystal
3 stages
1) ion exchange between the solution that washes the crystal - this is saliva and dental fluid with its hydration shell. It receives ions that neutralize the charge of the crystal: Ca, Sr, Co, PO, and citrate. Some ions can accumulate and also easily leave without penetrating into the crystal - these are K and Cl ions, other ions penetrate into the surface layer of the crystal - these are Na and F ions. The stage occurs quickly within a few minutes.
2) this is an ion exchange between the hydration shell and the surface of the crystal; an ion is separated from the crystal surface and replaced with other ions from the hydration shell. As a result, the surface charge of the crystal is reduced or neutralized and it becomes stable. Longer than stage 1. During few hours. Ca, F, Co, Sr, Na, P penetrate.
3) Penetration of ions from the surface into the crystal - called intracrystalline exchange, occurs very slowly and as the ion penetrates, the speed of this stage slows down. The ions Pa, F, Ca, Sr have this ability.
The presence of vacancies in the crystal lattice is an important factor in the activation of isomorphic substitutions within the crystal. The penetration of ions into the crystal depends on the R ion and the level of E it possesses; therefore, H ions, which are similar in structure to the H ion, penetrate more easily. The stage lasts for days, weeks, months. The composition of the HAp crystal and their properties are constantly changing and depend on the ionic composition of the liquid that washes the crystal and the composition of the hydration shell. These holy crystals make it possible to purposefully change the composition of hard tooth tissues under the influence of remineralizing solutions for the purpose of preventing or treating caries.
Organic substances of enamel
The share of organic matter 1 is 1.5%. In immature enamel up to 20%. Organic substances of enamel influence the biochemical and physical processes occurring in tooth enamel. Org.v-va nah-xia between apatite crystals in the form of bundles, plates or spirals. The main representatives are proteins, carbohydrates, lipids, nitrogen-containing substances (urea, peptides, cyclic AMP, cyclic amino acids).
Proteins and carbohydrates are part of the organic matrix. All remineralization processes occur on the basis of a protein matrix. Most of it is represented by collagen proteins. They have the ability to initiate remineralization.
1. a) enamel proteins – insoluble in acids, 0.9% EDTA. They belong to collagen- and ceramide-like proteins with a large amount of sulfur, hydroxyproline, gly, and lys. These proteins play a protective role in the process of demineralization. It is no coincidence that in the focus of demineralization at the stage of a white or pigmented spot, the number of these proteins is > 4 times. Therefore, a carious spot does not turn into a carious cavity for several years, and sometimes caries does not develop at all. In older people, caries > resistance. b) calcium-binding proteins of enamel. KSBE. They contain Ca ions in a neutral and slightly alkaline environment and promote the penetration of Ca from saliva into the tooth and back. Proteins A and B account for 0.9% of the total mass of enamel.
2. B. soluble in water, not associated with mineral substances. They do not have an affinity for the mineral components of enamel and cannot form complexes. There are 0.3% of such proteins.
3. Free peptides and individual amino acids, such as promine, gly, val, hydroxyproline, ser. Up to 0.1%
1) protective function. Proteins surround the crystal. Prevents the process of demineralization
2) proteins initiate mineralization. Actively participate in this process
3) provide mineral exchange in enamel and other hard tissues of the tooth.
Carbohydrates are represented by polysaccharides: glucose, galactose, fructose, glycogen. Disaccharides are in free form, and protein complexes are formed - phospho-glycoproteins.
There are very few lipids. Presented in the form of glycophospholipids. During the formation of the matrix, they act as connecting bridges between proteins and minerals.
Dentin is inferior in hardness. The most important elements of dentin are the ions Ca, PO, Co, Mg, F. Mg is 3 times more than in enamel. The concentration of Na and Cl increases in the inner layers of dentin.
The main substance of dentin consists of HAP. But unlike enamel, dentin is penetrated by a large number of dentinal tubules. Pain sensations are transmitted through nerve receptors. The dentinal tubules contain processes of odontoblast cells, pulp and dentinal fluid. Dentin makes up the bulk of the tooth, but is less mineralized than enamel; its structure resembles coarse-fibered bone, but is harder.
Organic matter
Proteins, lipids, carbohydrates, ...
Protein matrix of dentin - 20% of the total mass of dentin. Consists of collagen, it accounts for 35% of all organic dentin. This property is characteristic of lysine tissues of normal origin; it contains glucosaminoglycogens, galactose, hexasamites and heliuronic acids. Dentin is rich in active regulatory proteins that regulate the remineralization process. Such special proteins include amelogenins, enamelins, and phosphoproteins. Dentin, like enamel, is characterized by a slow exchange of minimal components, which is of great importance for maintaining tissue stability under conditions of increased risk of demineralization and stress.
Tooth cement
Covers the entire tooth with a thin layer. Primary cement is formed by a mineral substance in which collagen fibers and cellular elements - cementoblasts - pass in different directions. The cement of a mature tooth is little renewed. Composition: mineral components are mainly represented by Ca carbonates and phosphates. Cementum, like enamel and dentin, does not have its own blood vessels. In the apex of the tooth there is cellular cement, the main part is acellular cement. Cellular resembles bone, and acellular consists of collated fibers and an amorphous substance that glues these fibers together.
Dental pulp
This is loose connective tissue of the tooth, filling the coronal cavity and root canal of the tooth with a large number of nerves and blood vessels; the pulp contains collagen, but no elastic fibers; there are cellular elements represented by odontoblasts, macrophages and fibroblasts. The pulp is a biological barrier that protects the dental cavity and periodontium from infection, and performs a plastic and trophic function. It is characterized by increased activity of redox processes, and therefore high O consumption. Regulation of the energy balance of the pulp is carried out by coupling oxidation with phosphorylation. A high level of biological processes in the pulp is indicated by the presence of processes such as PPP, synthesis of RNA, proteins, therefore the pulp is rich in enzymes that carry out these processes, but carbohydrate metabolism is especially characteristic of the pulp. There are enzymes of glycolysis, TCA cycle, water-mineral metabolism (alkaline and acid phosphotose), transaminases, aminopeptidases.
As a result of these metabolic processes, many intermediate products are produced that come from the pulp into the hard tissues of the tooth. All this ensures a high level of ...., reactivity and protective mechanisms.
With pathology, the activity of these enzymes increases. With caries, destructive changes occur in odontoblasts, destruction of collagen fibers, hemorrhages appear, enzyme activity changes, and exchange of substances in the pulp changes.
Routes of entry of substances into hard tooth tissues and enamel permeability
The tooth has contact with mixed saliva, on the other hand - .... blood, the composition of the hard tissues of the tooth depends on their composition. The main part of the organic and mineral substances that enter the tooth enamel is contained in saliva. Saliva acts on tooth enamel and causes swelling or shrinkage of collagen barriers. As a result, a change in the permeability of the enamel occurs. Substances of saliva exchange with substances of enamel and the processes of de- and remineralization are based on this. Enamel is a semi-permeable membrane. It is easily permeable to H O, ions (phosphates, bicarbonates, chlorides, fluorides, cations Ca, Mg, K, Na, F, Ag, etc.). They determine the normal composition of tooth enamel. Permeability also depends on other factors: on the chemical structure of the substance and the strength of the ion. The sizes of apatites are from 0.13 - 0.20 nm, the distance between them is 0.25 nm. Any ions must penetrate the enamel, but determine the permeability with t.zr. Mr or ion sizes are not possible; there are other properties of the ion’s affinity for enamel hydroxyapatite.
The main route of entry of substances into the enamel is simple and facilitated diffusion.
The permeability of enamel depends on:
1) sizes of microspaces, filled. H O in the enamel structure
2) the size of the ion or the size of the molecule of the substance
3) the ability of these ions or molecules to bind to enamel components.
For example, the F ion (0.13 nm) easily penetrates the enamel and binds to the enamel elements in the damaged enamel layer, therefore it does not penetrate into the deeper layers. Ca (0.18 nm) – is adsorbed on the surface of enamel crystals, and also easily enters the crystal lattice, so Ca is deposited both in the surface layer and diffuses inside. J easily penetrate into the microspace of the enamel, but are not able to bind to HAP crystals, enter dentin, pulp, then into the blood and are deposited in the thyroid gland and adrenal glands.
The permeability of enamel decreases under the influence of chemicals. Factors: KCl, KNO, fluoride compounds. F interacts with HAp crystals, creating a barrier to the deep penetration of many ions and substances. The properties of the pron depend on the composition of the mixed saliva. Thus, secret saliva has different effects on the permeability of enamel. This is associated with the action of enzymes found in saliva. For example, hyaluronidosis > permeability of Ca and glycine, especially in the area of the carious spot. Chemotrypsin and whole phosphatose < CaF and lysine permeability. Acid phosphatosis > permeability to all ions and substances.
It has been proven that amino acids (lysine, glycine), glucose, fructose, galactose, urea, nicotinamide, vit, and hormones penetrate into tooth enamel.
Permeability depends on the age of the person: the greatest - after tooth eruption, it decreases by the time the tooth tissues mature and continues to decrease with age. From 25 to 28 years > resistance to caries, a complex exchange occurs while maintaining a constant composition of the enamel.
Saliva pH, as well as a decrease in pH under dental plaque, where organic acids are formed, permeability increases due to the activation of enamel demineralization by acids.
Caries > permeability. At the stage of white and pigmented spots > permeability, > the possibility of penetration of various ions and substances, as well as Ca and phosphates - these are compensatory reactions in response to active demineralization. Not every carious spot turns into a carious cavity; caries develops over a very long time.
Hyposalivation leads to the destruction of enamel. Caries that occurs at night is a nocturnal disease.
Surface formations on teeth
These are mucin, cuticle, pelicula, plaque, stone.
Mucin is a complex protein, related to salivary glycoproteins, which covers the surface of the tooth and performs a protective function, protects against mechanical and chemical influences, its protective role is explained by the characteristics, specificity of the amino acid composition and the characteristics of the content of sulfur, trianine, which contain up to 200 amino acids, pro... It is attached to sulfur and trianine residues through an O-glycosidic bond. N-acetylneuramine residues. to-you, N-acetylglucosamine, galactose and f..zy. The structure of the protein resembles a comb, which has ... proteins, residues consisting of amino acids, and carbohydrate components are arranged in protein chains, they are connected to each other by disulfide bridges and form large molecules capable of holding H O. They form a gel.
Pellicle
This is a thin, transparent film of carbohydrate-protein nature. Includes glycine, glycoproteins, some amino acids (ala, glu), Jg, A, G, M, amino sugars, which are formed as a result of the vital activity of bacteria. The structure contains 3 layers: 2 on the surface of the enamel, and the third in the surface layer of the enamel. The pellicle covers the dental plaque.
Plaque
White soft film located in the cervical area and on the entire surface. Removed during cleaning and hard food. This is a cariogenic factor. Represents a destructive organ with a large number of substances that are present in the oral cavity, as well as their waste products. 1 g of dental plaque contains 500 x 10 microbial cells (streptococci). There are early plaque (during the first day) and mature plaque (from 3 to 7 days).
3 hypotheses for plaque formation
1) …
2) precipitation of salivary glycoproteins that react in bacteria
3) pricipitation of intracellular polysaccharides. They are formed by streptococci, called dextran and levan. If you centrifuge dental plaque and pass it through a filter, two fractions are separated, cellular and acellular. Cellular – epithelial cells, streptococci, (15%). ....you, diphtheroids, staphylococci, yeast-like fungi - 75%.
In dental plaque, 20% is dry matter, 80% is H O. Dry matter contains minerals, proteins, carbohydrates, and lipids. From mineral ingredients: Ca – 5 mcg/per 1 g of dry plaque. P – 8.3, Na – 1.3, K – 4.2. There are microelements Ca, Str, Fe, Mg, F, Se. F soda in dental plaque in three forms:
1) CaF - Ca fluoride
1) CF protein complex
2) F in M/O structure
Some microelements reduce the susceptibility of teeth to caries F, Mg, others reduce resistance to caries - Se, Si. Proteins from dry plaque – 80%. The protein and amino acid composition is not identical to that of mixed saliva. As amino acids mature, they change. Gly, arg, lys, >glutomate disappears. Carbohydrates 14% - fructose, glucose, hexosamines, salic acids and acids, and glucosamines.
With the participation of enzymes from plaque bacteria, polymers are synthesized from glucose - dextran, and from fructose - levan. They form the basis of the organic matrix of dental plaque. The microorganisms involved in the pre...tion are split, respectively, by dext...heat and levanous cariogenic bacteria streptococci. Available in limited quantities: maktak, pyruvate, acetic acid, propionic acid, citric acid. This leads to a decrease in pH under dental plaque on the surface of the enamel to 4.0. These are cariogenic conditions. Therefore, dental plaque is one of the important etiological and pathogenic links in the development of caries and periodontal diseases.
Lipids
Early dental plaque contains triglycerides, glycerol, and glycerophospholipids. In a mature quantity < , complexes with carbohydrates are formed - glycerophospholipids.
Many hydrolytic and proteolytic enzymes. They act on the organic enamel matrix, destroying it. Relative glycosidoses. their activity is 10 times higher than in saliva. Acidic, alkaline phosphatases, pH, DN-noses. Peroxidases.
The metabolism of dental plaque depends on the nature of the microflora. If streptococci predominate in it, then pH<, but the pH of dental plaque can also increase due to the predominance of actives and staphylococci, which have urealytic activity, break down urea, NH, and deaminate amino acids. The resulting NH combines with the phosphates and carbonates of Ca and Mg and first amorphous carbonate and phosphate of Ca and Mg, non-crystalline HAP - -> crystalline, are formed.
Dental plaque mineralizes and turns into tartar. Especially with age, with certain types of pathology in children - tartar deposits are associated with congenital heart lesions, S.D.
Tartar (ZK)
This is a pathological discalcified formation on the surface of the teeth. There are supragingival, subgingival z.k. They differ in location, chemical composition and chemistry of formation.
Chemical composition of g.c.
Min. content 70 – 90% dry content.
Amount of mineral substances in s.c. various. Dark z.k. contains more minerals than light. What > zk is mineralized, mem > Mg, Si, Str, Al, Pb. First, the low-mineralized substances of ZK are collected, which are 50% composed of bruslit substances Ca NPO x 2H O.
Octocalcium phosphate CaH (PO) x 5H O
Carbonate apatites Ca (PO CO)
Ca (PO) CO (OH).
Hydroxyapatite Ca (PO) (OH
Viktolit – (Ca Mg) (PO)
Is in zk –F is contained in the same z-forms as in dental plaque.
Proteins, depending on the maturity of the cell, range from 0.1 to 2.5%. Number of proteins < as mineralization progresses. In the supragingival zone the soda content is 2.5%. In the dark supragingival zone – 0.5%, in the subgingival zone – 0.1%
Knowledge B. VZK are calcium-precipitating glyco- and phosphoproteins. The carbohydrate part of which is represented by galactose, fructose, ointment. In a ratio of 6:3:1.
The peculiarity of the amino acid composition is that there are no cyclic amino acids
GPL lipids are synthesized by dental plaque microorganisms. Capable of binding Ca to proteins and initiating the formation of HAP. There is ATP in the cell, it is both a source of energy and also a donor of organophosphorus. during the mineralization of brulite and its transformation into TAP. Brulite turns into octocalcium phosphate -> HAP (at pH>8). Brulite - ATP -> octocalcium phosphate -> HAP.
Biochemical changes in hard dental tissues during caries, prevention of caries by remineralization method
Initial biochemical changes occur at the boundary between the surface of the enamel and the base of the tartar. The primary clinical manifestation is the appearance of a carious spot (white or pigmented). In this area of enamel, demineralization processes first occur, especially pronounced in the subsurface layer of enamel, and then changes occur in the organic matrix, which leads to enamel permeability. Demineralization occurs only in the area of the carious spot and it is associated with an increase in the microspace between the HAP crystals, > the solubility of enamel in an acidic environment, 2 types of reactions are possible depending on the acidity:
Ca(PO)(OH) + 8H = 10Ca + 6 HPO + 2 HO
Ca(PO) (OH) + 2H = Ca(HO) (PO) (OH) + CA
Reaction No. 2 leads to the formation of apatite in the structure of which there are instead of 10.9 Ca atoms, i.e. < Ca/P ratio, which leads to the destruction of HAP crystals, i.e. to demineralization. You can stimulate the first type of reaction and inhibit demineralization. Stage 2 of caries development – the appearance of a caries plaque. This is a gel-like substance of carbohydrate-protein nature; microorganisms, carbohydrates, enzymes and toxins accumulate in it. The plaque is porous, carbohydrates easily penetrate through it. 3 floor – formation of organic acids from carbohydrates due to the action of enzymes of cariogenic bacteria. The pH shifts to the acidic side, destruction of enamel and dentin occurs, and the formation of a carious cavity.
Prevention and treatment of caries with remineralizing agents
Remineralization is a partial change or complete restoration of the mineral components of tooth enamel due to components of saliva or remineralizing solutions. Remineralization is based on the adsorption of minerals into carious areas. The criterion for the effectiveness of remineralizing solutions is such properties of enamel as permeability and solubility, disappearance or reduction of carious spots, < increase in caries. These functions are performed by saliva. Remineralizing solutions are used containing Ca, P, in the same proportions and quantities as in saliva, all the necessary microelements.
Remineralizing solutions have a greater effect than mixed saliva.
In saliva, Ca and P combine with organic complexes of saliva and the content of these complexes decreases in saliva. These solutions must contain F in the required amount, since it affects the rejuvenation of Ca and P in the hard tissues of the tooth and bone. At < concentration, HAP precipitates from saliva; in the absence of F, HAP does not precipitate, and octocalcium phosphate is formed instead of HAP. When there is a lot of F, instead of HAP, mineral substances that are unusual for these tissues and, more often, CaF.
Hypothesis of caries pathogenesis
There are several hypotheses:
1) neurotrophic caries is considered as a result of human conditions and the impact of environmental factors on him. The authors attached great importance to the central nervous system
2) trophic. The mechanism of caries development is a violation of the trophic role of odontoblasts
3) appeal theory. Caries is the result of pelation of enamel by complexes of mixed saliva. Caries is the result of simultaneous proteolysis of organs and mineralization of enamel
4) acidogenic or chemical-karyositotic. It is based on the effect of acid-reacting substances on tooth enamel and the participation of microorganisms in the carious process. It was proposed 80 years ago and forms the basis of the modern hypothesis of the pathogenesis of caries. Caries of decalcified tissues caused by acids, image. as a result of the action of microorganisms on carbohydrates.
Cariogenic factors are divided into general and local factors.
General:
include poor nutrition: excess carbohydrates, lack of Ca and P, deficiency of microelements, vitamins, proteins, etc.
Diseases and changes in the functional state of organs and tissues. Adverse effects during teething and maturation and in the first year after teething.
Electrical air (ionizing radiation, stress), which affects the salivary glands, the secreted saliva does not correspond to the normal composition, and it affects the teeth.
Local factors:
1) plaque and bacteria
2) changes in the composition and pH of mixed saliva (pH shift to the acidic side, lack of F, decrease in the amount and ratio of Ca and P, etc.)
3) carbohydrate diet, carbohydrate food residues.
Anti-cariogenic factors and dental caries resistance
1) susceptibility to caries depends on the type of mineralization of hard dental tissues. Yellow enamel is more caries-resistant. With age, the crystal lattice becomes denser and the caries resistance of teeth increases.
2) Caries resistance is promoted by the replacement of HAP with fluorapatites - stronger, more acid-resistant and poorly soluble. F is an anti-cariogenic factor
3) Caries resistance of the surface layer of enamel is explained by the increased content of microelements in it: stanium, Zn, Fe, Va, tungsten, etc., and Se, Si, Cd, Mg are cariogenic
4) Dental caries resistance is promoted by vit. D, C, A, B, etc.
5) Mixed saliva has anti-cariesogenic properties, i.e. its composition and properties.
6) Particular importance is attached to citric acid and citrate.
F and strontium
F is found in all tissues of the body. Available in several forms:
1) crystal. form of fluorapatite: teeth, bones
2) in combination with organic. in-you glycoproteins. Image of an organic matrix of enamel, dentin, bones
3) 2/3 of the total amount of F is in the ionic state in biol.
liquids: blood, saliva. A decrease in F in enamel and dentin is associated with a change in pit.H O.
It is easier for F to be included in the enamel structure in a slightly acidic environment, the amount of F in bones increases with age, and in the teeth of children it is found in increased quantities during the maturation of hard tooth tissues and immediately after eruption.
With very large amounts of F in the body, poisoning with fluorine compounds occurs. It is expressed in increased fragility of bones and their deformation due to disruption of R-Ca metabolism. As with rickets, but the use of vitamin D and A does not cause a significant effect on the disturbance of P-Ca metabolism.
A large amount of F has a toxic effect on the entire body, due to a pronounced inhibitory effect on the metabolic processes of carbohydrates, fats, and tissue respiration.
Role F
They take part in the process of mineralization of teeth and bones. The strength of fluorapatites is explained by:
1) amplification bonds between Ca ions in the crystal lattice
2) F binds to organic matrix proteins
3) F contributes to the formation of more durable crystals of HAP and F-apatites
4) F helps to activate the process of precipitation of apatites of mixed saliva and thereby increase. its remineralizing function
5) F affects the bacteria of the oral cavity, acid-forming properties are burned and thereby prevents the pH from shifting to the acidic side, because F inhibits ecolase and suppresses cliquelysis. The anti-caries effect of F. is based on this mechanism.
6) F takes part in regulating the entry of Ca into the hard tissues of the tooth, reducing the permeability of enamel to other substrates and increasing caries resistance.
7) F stimulates reparative processes in case of bone fractures.
 F reduces the content of radioactive strontium in the bones and teeth and reduces the severity of rickets. Sr competes with Ca for inclusion in the HAP crystal lattice, and F suppresses this competition.
F reduces the content of radioactive strontium in the bones and teeth and reduces the severity of rickets. Sr competes with Ca for inclusion in the HAP crystal lattice, and F suppresses this competition.
Ascorbic acid. Function. Role in the metabolism of tissues and organs of the oral cavity
1) the effect of the vitamin is associated with its participation in OM reactions. It accelerates the dehydrogenation of reduction. coenzymes NADH, etc., activates the oxidation of glucose by PFP, which is so characteristic of dental pulp.
2) Vitamin C affects the synthesis of glycogen, which is used in teeth as the main source of energy during the mineralization process.
3) Vit.C active. many enzymes of carbohydrate metabolism: in glycolysis - hexose, phosphofructokinose. In CGC...hydrogenosis. In tissue respiration - cytochrome oxidosis, as well as mineralization enzymes - alkaline phosphatosis
4) Vit.C is directly involved in the biosynthesis of protein, compound, procollagen in its transformation into collagen. This process is based on 2 reactions
proline - -axiproline
Ph-t: proline hydroxylase, cof-t: vit C.
Lysine – oxylysine f-t: lysine hydroxylase, cof-t: vit.C
Vitamin C performs another function: activation of enzymes by reducing disulfide bridges in enzyme proteins to sulhydryl groups. As a result of activation of alkaline phosphatosis, ... dehydrogenase, cytochromexidosis.
Vitamin C deficiency affects the condition of the periodontium, the formation of intercellular substance in the connective tissue decreases
5) vitamin deficiency changes the reactivity of tooth tissue. May cause scurvy.
Is it possible to regenerate enamel?
The tissue that forms enamel does not have the ability to self-heal and regenerate. Therefore, it is so important to closely monitor its condition and respond to any changes in a timely manner. However, today we have quite a wide range of opportunities to strengthen the upper protective layer on the teeth in case of thinning. We are talking about modern methods of remineralization and fluoridation.
The photo shows the procedure for fluoridation of teeth
This may be a one-time procedure or a therapeutic course prescribed by a doctor. In this case, the teeth are coated with special varnishes with a high concentration of fluoride and calcium. They help increase the resistance of hard tissues to external aggressive factors, and also reduce their sensitivity - eliminate hyperesthesia. By the way, such varnishes are also applied every time after professional cleaning of plaque and deposits in the dentist’s office. Remember that this procedure must be completed every six months for prevention.
But in case of noticeable damage to the enamel layer, it is necessary to correct the defect by filling with preliminary removal of the affected tissue. In another case, the installation of veneers or even dentures may be recommended if the destruction has severely damaged the outer coronal part.
The most popular manufacturers, review
The most effective enamel paints are products from well-known manufacturers. Among the best companies are the Japanese manufacturer Tamiya, the Irish brand HUMBROL, the English brand Dulux, the German company Caparol, the Danish company Sadolin and the German-Polish joint venture Hansa.
The advantage of these brands is explained simply; they have many years of experience in the production of paints and varnishes. All of the listed manufacturers have products of the highest quality and an excellent reputation.
Russian manufacturers are also actively working to create the highest quality products. The following companies can please customers with their results:
- Belgorod enterprise "Painting paints";
- Yaroslavl production "SpetsEmal";
- St. Petersburg.
The most effective enamel paints are products from well-known manufacturers.
All paints and varnishes have individual technical characteristics. Therefore, in order to choose the most optimal option, you need to familiarize yourself with all the subtleties and nuances of enamels.
Personal hygiene basics
Of course, it is impossible to insure against dental diseases, but we can reduce the risk of their occurrence to the very minimum. To do this, it is enough to ensure proper oral hygiene, that is, regularly brush your teeth day and evening, use preventive rinses and flosses after each meal, try to eat healthy foods, including those rich in calcium and phosphorus, and also avoid smoking and excessive consumption sweets, flour, carbonated drinks and alcohol.
Proper oral hygiene will help keep your teeth healthy
It is also important not to forget to visit the dentist every six months for preventive examinations and removal of deposits. It is equally important to promptly treat any pathological changes in the cavity and not delay a visit to a specialist when the first suspicious symptoms appear.
Important elements for strengthening dental tissues
It is with the right foods that our body receives useful substances, vitamins and microelements, which are most directly involved in maintaining the health of teeth and gums. Dental experts strongly recommend incorporating foods rich in the following into your daily diet:
- calcium, fluorine and vitamin D - these substances form the basis for the formation of bone tissue, including enamel,
- B vitamins – support gum health and strengthen the dentogingival ligamentous apparatus,
- Vitamin C is an important component for the health of the body as a whole. Its sufficient intake into the body along with food reduces the risk of bleeding gums and loosening of teeth, protects against the formation of ulcers and tissue infection,
- vitamin E – promotes the regeneration of damaged tissues, reduces the sensitivity of the mucous membrane and its susceptibility to mechanical irritants,
- vitamin A - a deficiency of this component disrupts salivation processes and the structure of the enamel layer, causing it to become rough and heterogeneous.
Vitamins are involved in maintaining healthy teeth and gums.
All these substances are extremely important for the health of not only the oral cavity, but also the entire body. Proper nutrition, a healthy lifestyle, as well as careful caring attitude towards your body - all this has the most positive effect on the appearance and condition of our teeth.
1Ippolitov, Yu. A. Functional morphology of human enamel, 2010.
Subtleties of selection and application
When choosing any paint and varnish product, you must carefully study the manufacturer’s recommendations written on the can and follow them. Thus, you should not use enamel intended for interior work when painting outside, as well as in the opposite case.
It is also necessary to pay attention to the paint container itself; there should be no damage to it, otherwise a broken seal of the can can lead to loss of properties of the product. In addition, the shelf life of the finishing material should not be expired, otherwise the enamel paint will lose its quality. Without fail, the container must have “GOST” on it, this will avoid counterfeiting.
Meanwhile, it is necessary to take into account the peculiarities of using paints and varnishes. As for enamels, despite the fact that there are a huge number of types combined into one group, each type has individual technical characteristics. Thus, nitrocellulose enamels are an ideal means for treating wooden bases, and alkyd paints are characterized by a wide range of uses, which includes not only outdoor work and indoor painting, but also decorative painting.
A smooth base without visible defects does not need putty before painting; you can limit yourself to cleaning and priming the surface. When working with enamel paint, it is permissible to use thinners and solvents of any type.
When working with enamel paint, it is permissible to use thinners and solvents of any type.